
Introduction
Primary packaging for pharmaceutical liquids covers every container that touches the drug directly — vials, ampoules, prefilled syringes, oral liquid bottles, and cartridges. These components serve as the critical barrier protecting drug safety and efficacy. According to FDA guidance, they must meet stringent requirements to prevent contamination, chemical interaction, and degradation throughout a product's shelf life.
Many pharmaceutical manufacturers still rely on legacy packaging formats that can no longer keep pace with modern drug formulations. Glass delamination has triggered multiple high-profile recalls — including Gilead's 2021 voluntary recall of Veklury (remdesivir) due to glass particulates. The rapid expansion of biologics and GLP-1 drugs is exposing compatibility gaps that traditional glass containers simply cannot address.
This article examines five packaging innovations reshaping how liquid pharmaceuticals are packaged, stored, and delivered, and why staying current on these advances helps pharmaceutical brands make smarter sourcing decisions.
TL;DR
- Advanced polymers replace glass vials, reducing delamination risk for biologics
- Blow-Fill-Seal technology delivers sterile liquid packaging in one automated step
- Smart packaging improves drug traceability and cold-chain monitoring
- Recyclable glass and bio-based materials gain ground as sustainability requirements tighten
- Prefilled syringes expand rapidly, driven by GLP-1 drug growth
Glass-to-Polymer Transitions — COP and COC Vials and Containers
Cyclic olefin polymer (COP) and cyclic olefin copolymer (COC) vials are displacing borosilicate glass for liquid pharmaceutical packaging — especially for biologics, vaccines, and high-pH formulations where glass delamination or adsorption can compromise drug integrity. Several high-profile recalls have accelerated the shift, pushing polymer alternatives from niche to mainstream consideration.
The Delamination Problem
Glass delamination is a specific failure mode where microscopic glass flakes shed into liquid drug product. The FDA issued an advisory in 2011 noting several drug recalls in 2010 and 2011 due to glass lamellae formation in small-volume glass vials. The problem has persisted: Hospira recalled Sodium Bicarbonate and Lidocaine HCl injections in October 2023, and Staska Pharmaceuticals recalled Ascorbic Acid Solution in October 2024 — both due to potential glass particulate matter.
These recalls have driven pharmaceutical companies to evaluate polymer alternatives that eliminate this risk entirely.
Functional Advantages of COP/COC for Liquid Drugs
COP and COC materials offer several performance benefits over traditional glass:
- Superior chemical inertness — no glass ion migration into drug product
- Lower protein adsorption — critical for biologic formulations where protein aggregation can reduce efficacy
- Break resistance — eliminates glass breakage risk during handling, shipping, and administration
- Freeze-thaw stability — larger coefficient of thermal expansion maintains container closure integrity at subzero and cryogenic temperatures better than glass
COP materials also deliver glass-like transparency (>91% light transmission) and low moisture permeability, making them suitable for light-sensitive and moisture-sensitive formulations.
Real-World Adoption
The prefilled syringe market's shift toward COP barrels illustrates this transition. Polymer prefilled syringes are projected to expand at an 11.06% CAGR through 2031, reaching 18% market share by 2025. Approximately 50% of pharmaceutical companies now use COP syringes for their ultra-low extractables profile and compatibility with sensitive biologic formulations.

The global cyclic olefin copolymer market was valued at $971 million in 2024 and is projected to reach $1.51 billion by 2033 at a 5% CAGR, driven largely by pharmaceutical packaging demand.
Strategic Implications
As biologics and GLP-1 drugs become a larger share of new drug approvals, liquid drug manufacturers face pressure to audit their primary packaging materials for compatibility. In 2024, 47% of FDA CDER novel drug approvals were biologics, up from 40.5% in 2022. For manufacturers whose pipelines skew toward injectables and high-sensitivity biologics, the compatibility audit isn't a future exercise — the data suggests it's overdue.
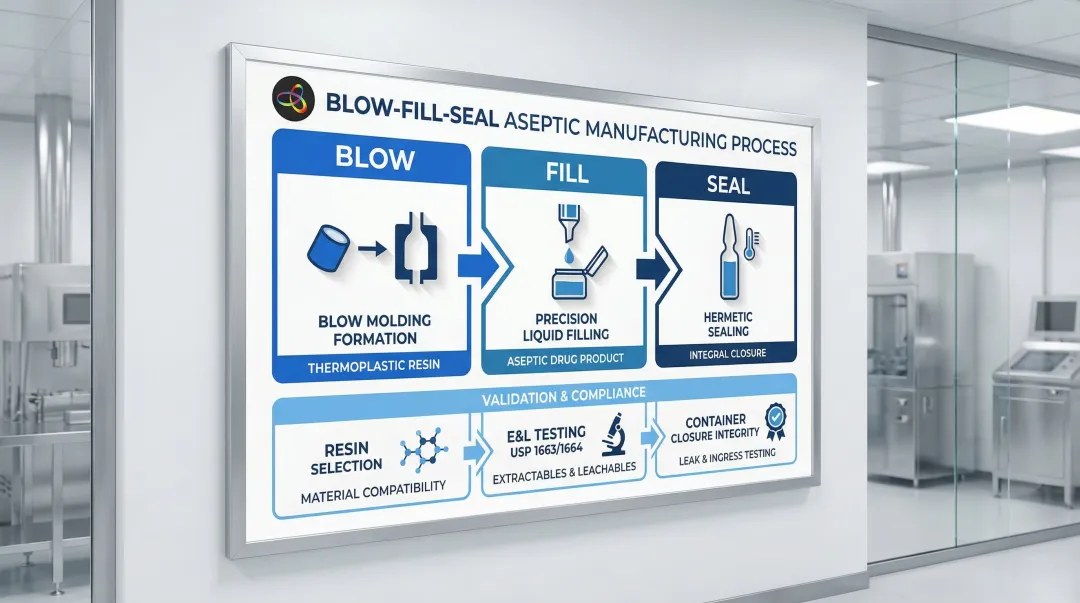
Blow-Fill-Seal (BFS) Technology for Sterile Liquid Packaging
Blow-Fill-Seal (BFS) is an advanced aseptic manufacturing process where the plastic container is formed, filled with sterile liquid drug product, and sealed in a single uninterrupted automated operation inside a controlled environment. This dramatically reduces contamination risk compared to traditional fill-and-finish methods.
How BFS Manifests in Pharmaceutical Liquids
BFS technology is widely used for:
- Ophthalmic solutions
- Inhalation liquids
- Oral unit-dose products
- Nasal sprays
- Parenteral solutions (increasingly)
All of these categories require high sterility assurance, making BFS an attractive alternative to conventional filling lines.
Why BFS Is Gaining Traction
The global BFS technology market is valued at $4.1 billion in 2026 and projected to reach $8.2 billion by 2036, reflecting a 7.2% CAGR. This growth is driven by several factors:
- Reduced human touchpoints: fewer operator interventions lower contamination risk in aseptic processing
- High-speed production: supports efficient, scalable manufacturing with minimal downtime
- Regulatory recognition: the FDA's 2004 guidance "Sterile Drug Products Produced by Aseptic Processing" explicitly acknowledges BFS as an automated process; EU GMP Annex 1 (2022 revision) details specific requirements for BFS equipment
A study of a closed robotic aseptic filling workcell across 260 production batches showed zero cases of microbial growth in over 250 media fills, with 99.3% of units meeting acceptance criteria — effectively eliminating operator intervention.
Material Compatibility Considerations
BFS uses thermoplastic materials, most commonly low-density polyethylene (LDPE) and polypropylene (PP). Compatibility with specific drug molecules must be validated through extractables and leachables (E&L) testing, governed by USP chapters <1663> and <1664>.
Key validation steps include:
- Selecting the appropriate resin based on drug formulation chemistry
- Running E&L studies to USP <1663>/<1664> standards
- Confirming container closure integrity under real-world storage conditions
Pharmaceutical companies adopting BFS should work with packaging partners experienced in thermoplastic compatibility to avoid late-stage reformulation surprises.
Smart Packaging and Digital Traceability for Liquid Pharmaceuticals
Smart packaging integrates digital technologies — including temperature sensors, NFC (Near Field Communication) chips, RFID tags, and QR/2D serialization codes — directly into or onto primary packaging for liquid pharmaceuticals, enabling real-time monitoring, authentication, and supply chain visibility.
Specific Applications for Liquid Drug Products
Cold-chain monitoring: Temperature and humidity sensors embedded in vials or bottles monitor cold-chain integrity for biologics and vaccines. CubeWorks offers the CubiSens XT1 NFC, a millimeter-scale wireless temperature tracker designed to attach to individual biopharma vials.
Authentication and dosing: NFC-enabled dropper bottles or prefilled syringes allow patients to scan packaging to verify authenticity and access dosing instructions. Ypsomed's SmartPilot, the first FDA-cleared connected add-on for autoinjectors (approved 2025), uses NFC labels to authenticate drugs and track injection data.
Regulatory Drivers
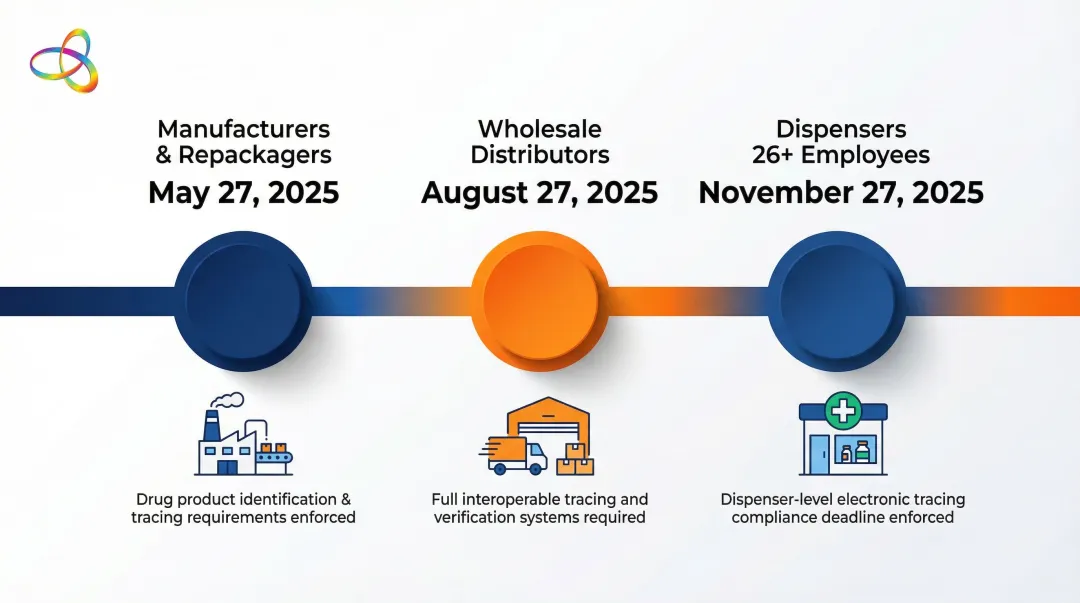
The U.S. Drug Supply Chain Security Act (DSCSA) requires full unit-level traceability across the pharmaceutical supply chain. The FDA's enforcement discretion periods set staggered compliance deadlines:
- Manufacturers and repackagers: May 27, 2025
- Wholesale distributors: August 27, 2025
- Dispensers (26+ employees): November 27, 2025
The EU Falsified Medicines Directive (Delegated Regulation 2016/161) has required a unique identifier encoded in a 2D Data Matrix barcode and an anti-tampering device on prescription medicinal products since February 9, 2019.
For pharmaceutical liquid packaging teams, this means serialization infrastructure — barcodes, tamper-evident features, and track-and-trace systems — must be built into primary packaging design from the start, not retrofitted later.
Patient Adherence Benefits
Smart connected packaging for liquid oral medications and self-injection devices can sync with mobile apps to send dose reminders, track usage history, and alert patients or caregivers to missed doses. This addresses a persistent gap: the WHO reports that in developed countries, adherence among patients with chronic diseases averages only 50%. Recent reviews show non-adherence rates among multi-morbid patients ranging from 44.1% to 76.5%.
Anti-Counterfeiting Impact
Counterfeit liquid pharmaceuticals — particularly injectable biologics and vaccines — pose serious patient safety risks globally. Holographic labels, invisible inks, and serialization codes make it far harder to replicate authentic packaging and enable point-of-dispensing verification.
That investment in authentication is paying off at scale: the global smart packaging market was valued at $28.5 billion in 2023 and is projected to reach $43.3 billion by 2030, growing at a 6.2% CAGR — driven in large part by pharmaceutical and healthcare adoption.
Sustainable and Eco-Friendly Primary Packaging for Liquid Drugs
The pharmaceutical liquid packaging industry is shifting toward materials and designs that reduce environmental impact — including high-PCR (post-consumer recycled) content glass bottles, recyclable mono-material plastic containers, bio-based barrier films for unit-dose pouches, and lightweighted designs that reduce material consumption per unit.
Regulatory Context
The EU's Packaging and Packaging Waste Regulation (Regulation EU 2025/40) entered into force and applies from August 12, 2026. While immediate packaging for medicinal products is explicitly exempt from recyclability requirements and minimum recycled content obligations to protect human health and safety, companies are still required to plan for Extended Producer Responsibility (EPR) compliance and labeling requirements.
Market Growth
The sustainable pharmaceutical packaging market was valued at $71.6 billion in 2022 and is projected to reach $146.3 billion by 2027, growing at a 15.4% CAGR. This significantly faster growth rate compared to the broader packaging market signals that sustainability is a commercial opportunity, not just a compliance burden.
Industry Examples
Several manufacturers are already translating this opportunity into tangible products:
- SGD Pharma introduced glass bottles with 20% post-consumer recycled (PCR) cullet at its Zhanjiang facility, becoming the first glass manufacturer in China to earn ISO 14021 certification for PCR glass packaging in 2024/2025.
- Corning's Viridian Vials reduce glass material by 20% and cut cradle-to-gate lifecycle emissions by up to 30% through lightweighting.
- SiO2 Medical Products uses plasma-enhanced chemical vapor deposition (PECVD) to apply a glass-like barrier coating to engineered polymers — combining the sustainability profile of plastic with the barrier performance of glass.

Patient-Centric Prefilled Delivery Systems for Liquid Pharmaceuticals
Prefilled syringes, dual-chamber cartridges, auto-injectors, and ready-to-use (RTU) vials represent a fundamental shift in how liquid drugs reach end users. These formats eliminate manual drawing-up steps and reduce dosing errors, making self-administration practical for patients managing conditions like diabetes, rheumatoid arthritis, and GLP-1-treated obesity.
GLP-1 Drug Surge
GLP-1 drugs like Ozempic and Wegovy have created massive demand for prefilled injection systems, as patients self-administer weekly injectable formulations at home. The market data reflects the scale of this shift:
- Eli Lilly's Mounjaro and Zepbound generated $39.5 billion in revenue in the first nine months of 2025
- Ypsomed reported 46.2% growth in autoinjector deliveries in H1 2025/26, heavily driven by GLP-1 demand
- Multi-dose pens and single-use auto-injectors are the dominant formats meeting this demand

The global prefilled syringes market is projected to reach $18.12 billion by 2033, growing at a 9.7% CAGR from 2026 to 2033.
Dual-Chamber Cartridges
While GLP-1 formats address stable liquid formulations, some biologics require a different approach entirely. Dual-chamber cartridges store a lyophilized (freeze-dried) drug and a liquid diluent separately in one container, reconstituting at the point of use — essential for biologics that degrade in solution form.
The FDA-approved Skytrofa (lonapegsomatropin-tcgd) uses a single-dose, dual-chamber prefilled cartridge with lyophilized powder and diluent. Edex (alprostadil) is also supplied in this format.
Error Reduction
A model-based economic evaluation in US healthcare settings showed that prefilled syringes reduced preventable adverse drug events from 1.39 per 100 administrations (vials) to 0.73 per 100 administrations — nearly a 50% reduction.
What's Driving These Innovations in Primary Liquid Packaging
Several converging forces — regulatory, technological, commercial, and environmental — explain why primary packaging for pharmaceutical liquids is evolving faster now than at any prior point in the past two decades.
Biologics and Specialty Drug Pipeline Growth
The surge in biologic drug approvals is pushing manufacturers to seek packaging formats specifically engineered for protein-based liquid formulations. These drugs are sensitive to:
- Light and UV exposure
- Oxygen and headspace gases
- pH shifts during storage
- Surface adsorption onto container walls
In 2024, 47% of FDA CDER novel drug approvals were biologics versus 53% small molecules. The FDA CDER reported 14,870 active INDs for drugs and non-biosimilar biologics in 2024, with non-biosimilar biologic INDs posting steady growth for four consecutive years.
Regulatory Compliance and Serialization Mandates
Global mandates from the FDA, EMA, and WHO around child-resistant closures, tamper evidence, and unit-level traceability are raising the technical bar for every primary liquid packaging solution. Companies unable to meet these standards face market access consequences.
Cost Efficiency and Manufacturing Automation
Pharmaceutical manufacturers are prioritizing packaging formats that integrate with automated aseptic fill-finish lines to reduce labor costs, minimize contamination risk, and increase throughput. Consolidated Design West supports this transition through end-to-end co-manufacturing services — from formulation through finished goods — with CGMP-compliant quality control built in at every stage.
Patient and Market Demand for Convenience
Direct-to-consumer and home healthcare trends are accelerating demand for liquid packaging formats that patients can safely and accurately use without clinical supervision — driving the premium on ergonomic, prefilled, and smart-enabled designs.
Future Signals for Primary Liquid Pharmaceutical Packaging
The next 1–3 years will be shaped by three converging developments: the maturation of BFS and polymer vial formats into mainstream use for biologics, growing regulatory clarity on digital labelling and QR-code-based product information, and the expansion of sustainability mandates to require disclosed recycled-content percentages and end-of-life labelling for pharma packaging. Companies that begin piloting these formats now will be better positioned to avoid costly compliance pivots later.
Technologies to Watch Closely
AI-assisted packaging design tools: Neural network models can now predict extractables and leachables risk before physical prototyping begins. A 2022 study in Environmental Science & Technology used support vector machine models to predict collision cross-section values from plastics with >93% accuracy — cutting the time and cost of drug-container compatibility testing.
Connected auto-injectors with IoT sensors: Ypsomed's YpsoMate On is a prefilled autoinjector with integrated connectivity that automatically logs injections via Bluetooth without requiring active pairing. Credence MedSystems' Connect Auto-Sensing Injection System incorporates automatic real-time monitoring of critical injection data. Both are moving from R&D to early commercial deployment.
For packaging teams, the practical implication is straightforward: format decisions made in the next 12–18 months will shape regulatory submissions, patient adherence data, and sustainability disclosures for years. Starting with one pilot — whether a BFS trial, a connected device proof-of-concept, or a recycled-content audit — is the lowest-risk way to build that readiness.
Frequently Asked Questions
What are the types of primary packaging in the pharmaceutical industry?
Primary packaging formats include vials, ampoules, prefilled syringes, bottles, blister packs, cartridges, and sachets. These are containers in direct contact with the drug product, each suited to different formulation types and routes of administration.
What is primary and secondary packaging in GMP?
In a GMP context, primary packaging directly contacts the product and must meet stringent sterility, compatibility, and barrier requirements, while secondary packaging provides outer protection and labeling. Both must be validated, documented, and controlled under GMP guidelines.
What are ideal requirements for pharmaceutical packaging?
Core requirements include chemical inertness (non-reactive with the drug), adequate barrier properties against moisture/light/oxygen, mechanical integrity, sterility where required, regulatory compliance (child resistance, tamper evidence, serialization), and patient usability.
What is the difference between glass and plastic primary packaging for pharmaceutical liquids?
Glass offers superior chemical inertness and barrier properties but carries risks of breakage and delamination for certain biologics. High-performance plastics like COP/COC offer break resistance and better protein compatibility. The choice depends on drug formulation, route of administration, and stability requirements.
What makes prefilled syringes a preferred primary packaging format for liquid drugs?
Prefilled syringes eliminate manual drawing-up errors, ensure accurate dose delivery, reduce contamination risk through a closed system, and support self-administration — making them preferred for biologics, vaccines, and increasingly for GLP-1 drug formulations.
How does smart packaging improve safety for pharmaceutical liquids?
Smart packaging embeds sensors or digital identifiers — temperature sensors, NFC/RFID, QR codes — into primary containers to monitor cold-chain integrity, verify authenticity, and connect patients to dosing apps. This reduces the risk of patients receiving compromised or counterfeit liquid drug products.


