
Introduction
Liquid products are unforgiving. The primary packaging must preserve product integrity, prevent leakage, enable precise dispensing, and satisfy growing consumer demand for sustainability—before a single secondary layer is added. For brands in beauty, wellness, food and beverage, and pharmaceuticals, choosing the right format is one of the most consequential decisions in product development.
The stakes are high. Label errors and packaging mishaps cost the food industry an estimated $1.92 billion in direct recall expenses in 2024, averaging $10 million per event. For liquids specifically, packaging failures (oxidation, contamination, seal integrity failures) can damage both brand reputation and product efficacy. In pharmaceuticals, seal failures accounted for approximately 1–4% of all FDA recalled product IDs annually between 2019 and 2025.
This guide covers the most innovative packaging formats for liquids, advanced dispensing technologies, sustainable material options, industry-specific considerations, and a practical framework for choosing the right solution for your product.
TLDR:
- Airless packaging offers 95-100% product evacuation and extends shelf life 2-3x by eliminating oxygen contact
- Flexible pouches reduce material usage by up to 80% versus rigid bottles while maintaining barrier protection
- Switching to PCR plastics, refillable systems, or mono-material designs cuts packaging waste with measurable cost ROI
- Dispensing technology—airless pumps, bag-on-valve, precision droppers—directly impacts user experience and product performance
- Evaluate formulation compatibility, regulatory compliance, and production scalability before finalizing any primary packaging format
What Is Primary Packaging for Liquid Products and Why Does It Matter
Primary packaging is the first layer of material in direct contact with your liquid product: the bottle, pouch, vial, tube, or carton that holds the formulation. It differs from secondary packaging (the outer box) and tertiary packaging (shipping containers) in one important way—it carries the highest functional responsibility.
That means protecting the liquid from degradation, preventing leaks, ensuring material compatibility, and delivering a dispensing experience that feels intuitive to the end user.
Liquids pose unique challenges compared to solids or powders. They require:
- Barrier protection against oxygen and light that can degrade active ingredients
- Leak-proof sealing to prevent product loss and contamination
- Material compatibility to avoid chemical reactions between packaging and formulation—critical for reactive ingredients in serums, pharmaceuticals, or acidic beverages
- Precise dispensing that controls dose, prevents waste, and works naturally for the consumer
These requirements explain why this category commands serious investment. The global liquid packaging market was valued at $371.44 billion in 2024 and is projected to reach $499.2 billion by 2030, growing at a 5.1% CAGR—driven by rising demand for convenient, sustainable, and lightweight solutions across food and beverage, pharmaceutical, and personal care industries.
Top Innovative Primary Packaging Formats for Liquid Products
Airless Packaging Systems
Airless packaging—typically pump-based bottles or jars with a rising base plate—eliminates air contact with the liquid by using a vacuum-sealed piston mechanism that pushes product upward as you dispense. That makes these systems a practical choice for oxidation-sensitive products like vitamin C serums, retinol formulations, and liquid pharmaceuticals.
Key benefits:
- Achieves 95-100% product evacuation, compared to 85-90% for traditional dropper bottles
- Prevents oxidation, extending product stability by 2-3 times over standard packaging
- Supports preservative-free formulations by minimizing microbial contamination risk

The global airless packaging market was valued at $6.69 billion in 2024 and is expected to grow at a 6.1% CAGR through 2030. Airless systems are gaining traction in clean beauty specifically because they align with consumer demand for formulations free from synthetic preservatives.
Flexible Pouches and Stand-Up Formats
Flexible stand-up pouches have evolved from beverage and baby food applications into premium beauty oils, liquid supplements, and household products. Innovations include spout pouches, re-sealable closures, and multi-layer barrier films that extend shelf life while significantly cutting material weight.
Material efficiency:
- Flexible pouches reduce overall material usage by up to 80% compared to rigid bottles
- Choosing inverted pouches over rigid plastic bottles results in 63% less plastic, 61% less fossil fuel consumption, and 76% less water usage
The global stand-up pouches market was valued at $14.42 billion in 2025 and is estimated to grow at a 5.29% CAGR to reach $19.76 billion by 2031, driven by e-commerce durability and significant sustainability gains.
Premium examples:
- Bark Bistro launched a 4-oz resealable spouted pouch for Buddy Butter using PET outer print web, nylon oxygen barrier, and polyethylene sealant layers
- Prestone debuted a self-standing pouch with child-resistant spout using 60% less plastic than traditional formats
For brands where dosing precision and contamination control are non-negotiable, unit-dose formats take a different approach.
Unit-Dose and Single-Use Formats
Unit-dose packaging—ampoules, stick packs, single-use vials—reduces contamination risk, delivers exact dosing, and raises perceived product value across pharmaceutical and cosmetic applications.
The global unit dose packaging market was valued at $29.1 billion in 2024 and is projected to reach $43.8 billion by 2029 (8.5% CAGR). The liquid segment accounts for 42.5% market share, widely used for injectable drugs, vaccines, and cosmetic serums.
Applications:
- Single-application facial serums for freshness and exact dosing
- Travel-size liquid supplements
- Dosage-controlled pharmaceutical liquids
Ritedose Corp. introduced the SuredoseRx dispensing device—a single-dose, blow-fill-seal ampoule for liquid medications that replaces foil-lidded cups to reduce spillage and contamination.
Glass and Aluminum Bottles and Vials
Glass and aluminum remain highly relevant for premium and pharmaceutical liquids, offering distinct advantages:
Glass:
- Chemical inertness: No leaching or interaction with reactive formulations (validated by USP <660> testing standards)
- Premium positioning: High-end aesthetic for cosmetics, beverages, and perfumes
- Design innovations: Specialty coatings, embossing, and custom shapes enable brand differentiation
Aluminum:
- Infinite recyclability: Aluminum boasts a 75% global recycling rate, outperforming PET (47%) and glass (42%)
- Light-blocking properties: Protects photosensitive formulations from UV degradation
- Energy efficiency: Recycling requires 95% less energy than primary production

For brands navigating both regulatory requirements and sustainability commitments, glass and aluminum frequently outperform plastic alternatives on barrier performance, recyclability, and consumer perception.
Advanced Dispensing Technologies Transforming Liquid Packaging
Dispensing Technologies for Liquid Packaging
Airless and Vacuum Pump Dispensers
Airless pumps eliminate oxidation at the point of use and deliver consistent dose quantities. Compared to traditional open jars or non-vacuum pumps, they improve product utilization rates — making them essential for skincare actives (retinol, vitamin C), pharmaceutical liquids, and personal care products where formulation stability directly impacts efficacy.
Precision Dropper and Pipette Systems
Calibrated droppers for serums, CBD oils, and liquid supplements provide exact dosing and user control. Modern innovations include:
- Tamper-evident closures (required by FDA 21 CFR 211.132 for OTC liquids)
- Child-resistant closures (mandated by 16 CFR 1700.14 for specific liquid OTCs with iron content ≥250 mg)
- Single-dose dropper systems that prevent contamination
The global dropper system market is estimated at $72.9 billion in 2025 and forecast to reach $121.1 billion by 2035 (5.2% CAGR), according to industry market forecasts.
Spray and Fine-Mist Dispensers
Bag-on-valve (BOV) technology represents a major dispensing innovation. BOV systems keep the liquid product completely separated from eco-friendly propellants (nitrogen or compressed air), offering:
- 99-100% product evacuation with zero waste
- 360-degree spray angles for consistent application
- Zero propellant contamination of the product
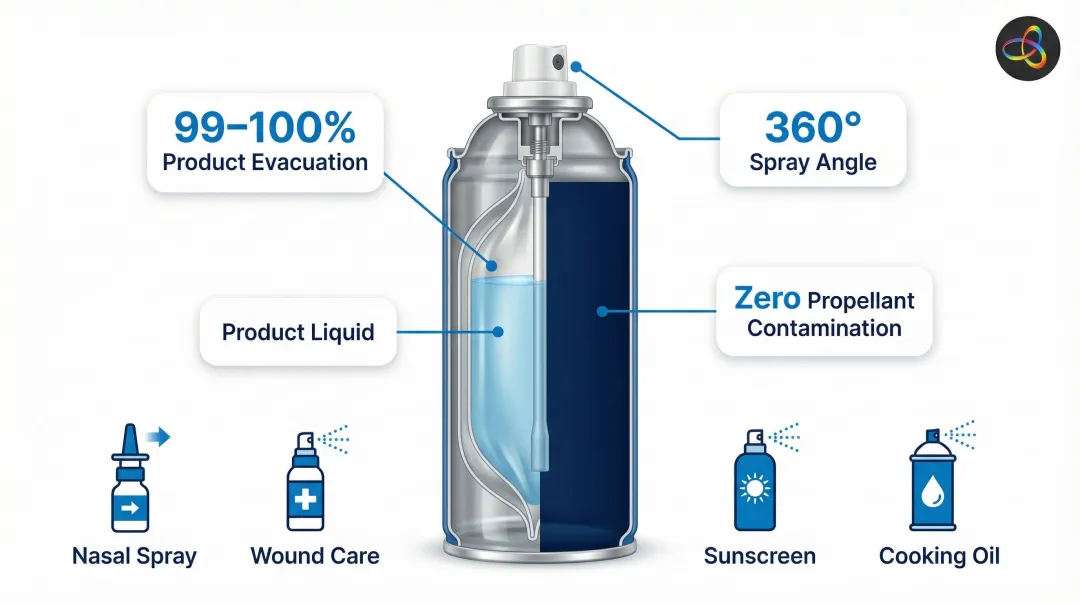
BOV is used for saline nasal sprays, wound care products, sun care lotions, and cooking oils.
Measuring Caps and Dose-Controlled Closures
Common in liquid detergents, cough syrups, and nutraceuticals, measuring caps reduce over-use, improve consumer experience, and support accurate dosing claims. Tamper-evident and child-resistant versions are available for products subject to FDA or CPSC safety requirements — a critical consideration when selecting closures for liquid OTC or household chemical formats.
Sustainable Primary Packaging Solutions for Liquid Products
PCR Plastics and Recycled Content
Post-consumer recycled (PCR) plastic is one of the most commercially accessible sustainable options for liquid brands. PCR content reduces reliance on virgin petroleum-based plastics while maintaining performance.
Current adoption:
- PCR usage in skincare containers increased by 22% since 2020
- HDPE bottles are available with 30% PCR content (options up to 100% PCR)
- Plastic tubes with sleeves can contain up to 60% recycled material
Regulatory compliance:
Compliance requirements differ by market:
- US: The FDA evaluates recycled plastics in food packaging case-by-case to ensure contaminants don't migrate (Guidance for Industry: Use of Recycled Plastics in Food Packaging)
- EU: EFSA assesses plastic recycling processes under Commission Regulation (EU) 2022/1616
Refillable and Reusable Systems
Refillable primary packaging has seen measurable adoption in premium beauty, household cleaning, and personal care. The model pairs a durable primary container with lightweight refill pouches or cartridges.
Brand benefits:
- Reduced packaging waste per unit
- Increased customer loyalty through repeat purchase mechanics
- Strong sustainability story that resonates with consumers
Unilever ran over 50 refill and reuse pilot projects globally. In Indonesia, community refill stations reached over 6,000 customers in 2023, saving approximately 6 tonnes of plastic through 91,000 litres of products sold.
Bioplastics and Plant-Based Materials
Bioplastics derived from sugarcane, cornstarch, or algae present real opportunities — but the terminology matters. Three terms are often used interchangeably when they aren't:
- Bio-based: Made from plants but not necessarily biodegradable
- Biodegradable: Microorganisms convert material into water, CO₂, and biomass under specific conditions
- Compostable: Designed for composting in municipal or industrial facilities (defined by ASTM D6400 and ISO 17088)

The bioplastic packaging market is expected to grow from $6.27 billion in 2025 to $18.89 billion by 2031 (20.18% CAGR). That growth comes with accountability — claims must match local recycling and composting infrastructure, or brands risk greenwashing exposure under the FTC Green Guides and EU Green Claims Directive.
Lightweight and Mono-Material Design
Lightweighting (reducing wall thickness) and mono-material design (using a single material type for bottle, cap, and pump) reduce overall material consumption and make entire packs recyclable as a single stream.
Carbon savings example:
Coca-Cola reduced PET bottle weight from 21g to 18.5g — a 12% reduction expected to cut PET use by 3 megatons by 2025.
Mono-material construction — full PE or full PP laminates — works because all layers share the same plastic type. That makes sorting easier and produces cleaner, more efficient recycling streams.
The 7 R's Framework:
Brands can evaluate their liquid packaging strategy using the 7 R's — Rethink, Refuse, Reduce, Reuse, Recycle, Recover, Re-educate — a hierarchy of sustainable actions from design intent to end-of-life consumer education.
Industry-Specific Packaging Considerations for Liquid Products
Beauty and Personal Care (Serums, Oils, Toners, Shampoos)
Material compatibility with active ingredients is paramount. High-potency actives require inert, light-blocking, and air-restrictive primary packaging:
| Active Ingredient | Packaging Interaction Risk | Recommended Solution |
|---|---|---|
| Vitamin C | Oxidizes rapidly with air and UV exposure | Airless pumps; amber or opaque glass |
| Retinol | Photodegradation from UV light | Airless technology; dark glass or HDPE |
| AHAs/Glycolic Acid | Low pH can corrode certain plastics | Glass or lined polymers |
Pump precision and premium unboxing aesthetics are primary drivers. Innovations like airless systems and frosted glass vials address both functional and brand positioning needs simultaneously.
Food, Beverage, and Nutraceutical Brands
Food and beverage packaging operates under strict federal oversight. The FDA regulates food contact substances under 21 CFR 170-186, requiring each material to be authorized via regulation, GRAS status, prior sanction, or an effective Food Contact Substance Notification (FCN).
Beyond compliance, brands must address three core functional requirements:
- Barrier protection against oxygen and UV to preserve shelf life
- Tamper-evidence features to meet retailer and regulatory standards
- Aseptic filling compatibility for sterile, shelf-stable formats
The aseptic packaging market is forecast to reach $141.06 billion by 2031 at a 10.31% CAGR. Multi-layer barrier cartons and flexible pouches address these needs at scale.
Pharmaceutical and Wellness Liquid Products
Liquid pharmaceutical and wellness products face the most demanding packaging requirements of any category. Under 21 CFR 211.94, container closure systems must protect against external factors that cause deterioration or contamination. USP Chapter <1207> outlines deterministic testing methods — laser-based gas headspace analysis and vacuum decay — to verify container closure integrity, sterility, and leak prevention.
Compliance obligations extend to consumer safety as well. Child-resistant closures are mandatory for specific liquid OTCs under the Poison Prevention Packaging Act (16 CFR 1700.14). Unit-dose formats are increasingly preferred for both regulatory alignment and patient convenience.
How to Choose the Right Primary Packaging for Your Liquid Product
Evaluate these four criteria sequentially:
1. Formulation Compatibility
Does the packaging material react with or degrade your liquid's active ingredients? Test for chemical interactions, pH compatibility, and potential leaching. For reactive formulations (acids, retinoids, essential oils), glass or specific lined polymers are often required.
2. Functional Performance
Does the dispensing mechanism deliver the right dose with the right user experience? Consider viscosity, required dose accuracy, and target consumer expectations. Airless pumps suit high-value serums; measuring caps work for liquid detergents; BOV dispensers excel for mists and sprays.
3. Sustainability Alignment
Does the format support your brand's environmental commitments and target market's values? According to Euromonitor's 2025 survey, 53% of global consumers trust packaging labels for sustainability claims, with recyclability the most trusted claim (57%). Evaluate PCR content, refillability, recyclability, and lifecycle impact.
4. Production Scalability
Can the chosen packaging be filled efficiently at the required volume on available or accessible filling lines? Startups and established brands alike often underestimate this step, particularly the interaction between formulation chemistry and packaging material.

Working with an experienced packaging partner early in development prevents costly reformulations or line changeovers later. Consolidated Design West has spent over 34 years helping brands in beauty, wellness, food and beverage, and pharmaceuticals navigate these decisions, from format selection and material sourcing through to finished goods production via their co-manufacturing partnership with Respect Manufacturing.
Frequently Asked Questions
What is primary packaging for liquid products?
Primary packaging is the first material layer in direct contact with the liquid—the bottle, pouch, vial, or tube responsible for product protection, integrity, and dispensing. It's distinct from secondary packaging (outer box) and tertiary packaging (shipping containers).
What are the latest packaging innovations for liquid products?
Current innovations include airless pump systems for oxidation-sensitive formulations, bag-on-valve dispensers offering 360-degree spraying, flexible stand-up pouches with barrier films reducing material by 80%, unit-dose ampoules for precise dosing, PCR and bioplastic materials, and smart packaging with embedded freshness sensors or QR codes.
What is the most eco-friendly packaging for liquids?
Infinitely recyclable materials like glass and aluminum have the strongest environmental credentials, with aluminum achieving a 75% global recycling rate. PCR plastics and refillable systems follow closely. The "most eco-friendly" option depends on product type, local recycling infrastructure, and full lifecycle impact.
What are the 7 R's of sustainable packaging?
The 7 R's form a hierarchy that moves from upstream decisions to end-of-life responsibility: Rethink the packaging concept, Refuse unnecessary materials, Reduce what remains, Reuse where possible, Recycle what can't be reused, Recover energy from the rest, and Re-educate consumers on proper disposal. Applied in order, they push brands toward reduction first—not just recyclability.
What dispensing options are available for liquid primary packaging?
Main options include airless pumps (for oxidation-sensitive products), standard pumps, precision droppers (for serums and oils), spray/mist dispensers including bag-on-valve technology, measuring caps (for detergents and syrups), and dose-controlled closures. Choice depends on product viscosity, required dose accuracy, and target consumer experience.
How do I choose the right primary packaging for my liquid product?
Four criteria should drive the decision:
- Formulation compatibility — does the packaging react with active ingredients?
- Dispensing functionality — does it deliver the right dose and user experience?
- Sustainability goals — does it align with your brand's environmental commitments?
- Production scalability — can it be filled efficiently at your required volumes?
Working with a packaging partner who can evaluate all four at once prevents costly redesigns later.


