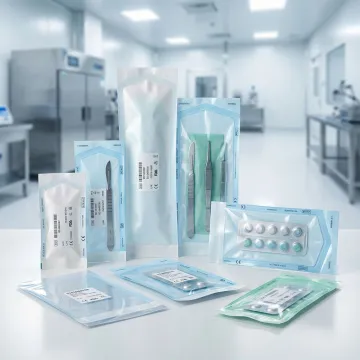
What materials are used in medical pouch packaging?
Medical pouch packaging utilizes specialized materials including medical-grade plastics (PET, HDPE, LDPE), aluminum foil laminates, and Tyvek for sterile barrier systems. Material selection depends on your specific requirements for barrier protection, sterilization method compatibility (autoclave, ethylene oxide, gamma radiation), and regulatory standards. We work with FDA-compliant materials that provide moisture barriers, oxygen protection, and puncture resistance while maintaining sterility throughout the product lifecycle. Our materials engineering team ensures optimal material selection for your medical device or pharmaceutical product's unique needs.
How do you ensure regulatory compliance for healthcare pouches?
Our comprehensive compliance program addresses FDA regulations, ISO 11607 standards for medical packaging, and international requirements including EU MDR. We maintain detailed documentation systems tracking material certifications, validation protocols, and manufacturing records. Our quality control processes include sterility testing, seal integrity validation, biocompatibility verification, and accelerated aging studies. Every production run undergoes rigorous inspection with full lot traceability. Our experienced regulatory team stays current with evolving healthcare standards, ensuring your medical pouches meet all applicable requirements for your target markets and distribution channels.
What types of medical products can be packaged in pouches?
Our medical pouch packaging accommodates a wide range of healthcare products including surgical instruments, diagnostic devices, catheters, syringes, wound care products, pharmaceutical tablets and capsules, sterile components, implantable devices, and single-use medical tools. We engineer custom pouch configurations for various product sizes, shapes, and sterilization requirements. Whether you need individual device packaging, combination product pouches, or pharmaceutical unit-dose packaging, our flexible manufacturing capabilities and material options can be tailored to protect your specific medical product while maintaining sterility and ensuring easy, contamination-free access.
What sterilization methods are compatible with your medical pouches?
Our medical pouch packaging is engineered for compatibility with all major sterilization methods including steam autoclave, ethylene oxide (EtO) gas, gamma radiation, electron beam (E-beam), and hydrogen peroxide plasma sterilization. Material selection and pouch construction are optimized based on your chosen sterilization protocol to ensure material integrity, seal strength, and sterile barrier maintenance post-sterilization. We conduct validation testing to confirm sterilant penetration, material compatibility, and package performance. Our technical team provides guidance on optimal sterilization methods for your specific product and packaging configuration, ensuring reliable sterility assurance.
How long does medical pouch packaging production take?
Medical pouch packaging timelines vary based on project complexity, customization requirements, and regulatory validation needs. Standard medical pouches with existing tooling typically require 3-4 weeks from order to delivery. Custom-designed healthcare pouches involving new tooling, material development, or extensive compliance testing may require 8-12 weeks for the complete development and validation process. Our project management team provides detailed timelines during consultation, accounting for design approval, material sourcing, manufacturing, quality testing, and regulatory documentation. For established products with validated packaging, we offer expedited production schedules and maintain safety stock programs to ensure consistent supply.
Do you provide packaging design services for medical pouches?
Yes, our comprehensive design services cover all aspects of medical pouch packaging development. Our team creates custom packaging solutions optimized for your product protection, sterilization requirements, user convenience, and regulatory compliance. Services include material selection, pouch configuration design, seal pattern engineering, opening feature development, printing layout with regulatory labeling, and usability testing. We utilize CAD modeling and prototyping to validate designs before production. Our designers collaborate closely with your team, regulatory consultants, and end-users to ensure the final packaging meets functional requirements, enhances product presentation, and facilitates safe, intuitive use in clinical settings.
What minimum order quantities do you require for healthcare pouches?
Minimum order quantities for medical pouch packaging vary based on customization level, materials, and printing requirements. Standard medical pouches with existing tooling typically have minimums of 5,000-10,000 units. Fully customized healthcare pouches with specialized materials, unique configurations, or complex printing may require minimums of 25,000-50,000 units to optimize tooling costs and ensure economic production. For startups and small-volume medical device manufacturers, we offer flexible solutions and can discuss phased production approaches. Our sales team works with you to balance your inventory needs, budget constraints, and quality requirements while optimizing total cost of ownership.
Can you handle fulfillment and distribution for medical packaging?
Yes, our integrated supply chain services include warehousing, fulfillment, and distribution specifically designed for medical packaging products. We maintain temperature-controlled, secure storage facilities with lot segregation and first-in-first-out inventory management. Our fulfillment services handle pick-and-pack operations, just-in-time delivery schedules, and direct-to-facility shipping. We provide comprehensive inventory tracking, automated reorder notifications, and detailed shipping documentation to support your supply chain compliance requirements. This turnkey approach eliminates the need for multiple vendors, reduces handling risks, and ensures your medical pouches are consistently available when and where you need them for production or distribution.